Medical Device CNC Machining
Built for compliance-driven builds: medical device CNC machining, precision machining for surgical instruments, and titanium Ti-6Al-4V ELI implant machining—with DFM-first collaboration, traceability-ready documentation, and inspection outputs (FAI/CMM)
STEP / IGES / SLDPRT / PDF accepted
- ±0.00019" tol. • Titanium • Magnesium • 5-axis CNC • ISO 9001

ISO 13485

Material traceability
CMM reporting

Revision Control
Why CNC Machining Powers Medical Devices
Medical devices reward repeatability, traceability, and clean interfaces. CNC machining turns regulatory intent—risk controls, verified CTQs, and documented inspection—into buildable geometry for instruments, implants, diagnostics, and device housings.
Burr and edge control for patient-facing parts
For surgical instruments and implant-adjacent components, burrs and sharp edges become usability, debris, and cleanability risks. DFM should define edge breaks, deburr method, and verification points.
Materials + finishes that survive sterilization
Common builds use medical-grade stainless steels, titanium, and polymers like PEEK. Finish selection (e.g., electropolish/passivation) is often driven by corrosion resistance and reprocessing cycles.
Audit-ready documentation and revision discipline
A medical device supply chain needs controlled revisions, traceability (material certs/lot control), and inspection outputs (FAI/CMM) to support validation and change management.
| Equipment interface | What CNC enables | What to specify on drawings | Common failure mode |
| Instrument hinge / jaw interfaces | Repeatable profiles + controlled radii | Datums, GD&T, edge break, surface finish Ra | Burrs, binding, inconsistent actuation |
| Implant threads / mating surfaces | Stable fits in titanium/316L and clean finishes | Thread class, concentricity, finish, cleaning notes | Galling, poor assembly, cosmetic rejects |
| IVD manifolds / housings | Leak-resistant interfaces + inspectable geometry | Flatness, sealing surfaces, port patterns, cleanliness | Leaks, contamination, rework loops |
Engineering Pain Points We Solve for Medical Devices
Medical device teams balance performance, manufacturability, and compliance. The fastest programs prevent late surprises by defining CTQs early (tolerances, finishes, edge conditions), aligning inspection method, and keeping revision control tight from prototype to production.
Burrs, debris, and cleanability risk
Burrs, sharp edges, and crevices complicate cleaning and can create functional or cosmetic failures—especially on reusable instruments.
Tolerance stack-up in small assemblies
Micro features, mating fits, and tight GD&T need clear datums and realistic tolerance strategy to avoid rework loops.
Documentation expectations
Supplier qualification often requires material certs, traceability, and inspection outputs (FAI/CMM) aligned to your CTQs.
Our CNC Machining Capabilities for Medical Devices
Milling for housings, manifolds, and fixtures
Best for diagnostics/IVD housings and microfluidic manifolds where sealing faces, flatness, and port patterns drive performance and cleanability.
Turning / Swiss for micro-components
Swiss machining medical components is common for pins, sleeves, shafts, and complex turned parts that need repeatable diameters and controlled burrs.
Finishing + edge conditioning
Electropolishing and passivation for medical parts, bead blasting, anodizing (housings/fixtures), and controlled deburr strategies—planned from DFM.
| Capability area | Typical battery-line parts | CTQ features we ask you to highlight | Helpful notes |
| Precision milling | IVD housings, microfluidic manifolds, instrument handles | Datums, sealing faces, flatness, Ra, port patterns | Add cleanability + reprocessing notes where relevant. |
| Swiss / turning | Pins, sleeves, shafts, fasteners, small connectors | Edge break, runout, thread class, concentricity | Good for high-mix micro parts and repeatability. |
| Implant machining | Plates, screws, spacers in titanium/316L | Threads, surface finish, cosmetic zones, cleaning notes | Plan for galling risk and finish compatibility. |
| Inspection outputs | FAI packages, CMM reports, material certs | CTQ list + method (CMM, gage, optical) | Supports traceability and validation documentation. |
What we machine for Medical Devices
Built to match engineering searches like medical device CNC machining and precision machining for surgical instruments—plus regulated documentation needs like FAI CMM inspection for medical device parts.

Surgical instruments & mechanisms
Focus: burr control, repeatable hinge/jaw geometry, surface finish Ra, and inspection for critical fits.

Implant & orthopedic components
Supports titanium Ti-6Al-4V ELI implant machining and medical-grade stainless components where fit, threads, and finish drive outcomes.

Diagnostics / IVD housings & manifolds
Sealing faces, port patterns, cleanability notes, and tight tolerance interfaces—optimized with DFM-first iteration.
| Subsystem | Common machined parts | What to specify | Risk if missed |
| Surgical instruments | Jaws, hinges, handles, guides, pivots | Datums, edge condition, Ra, hardness notes | Burrs, binding, inconsistent actuation |
| Implants / ortho | Plates, screws, spacers, trial components | Threads, surface finish, cosmetic zones, cleaning notes | Galling, fit issues, cosmetic rejects |
| Diagnostics / IVD | Housings, microfluidic manifolds, frames | Flatness, sealing surfaces, port patterns, cleanliness | Leaks, con |
FDA QMSR (21 CFR 820), ISO 13485, ISO 14971 risk management, GD&T, surface finish Ra, traceability, FAI/CMM.
Medical Device procurement workflow (DFM → Inspection → Handoff)
A practical sequence for medical device OEMs and contract manufacturers. It reduces ambiguity for CTQs like edge condition, surface finish Ra, and datum-driven fits—while keeping revisions and documentation audit-ready.
Robotics & automation CNC machining workflow — from CAD upload to production handoff (fixture + QC plan).
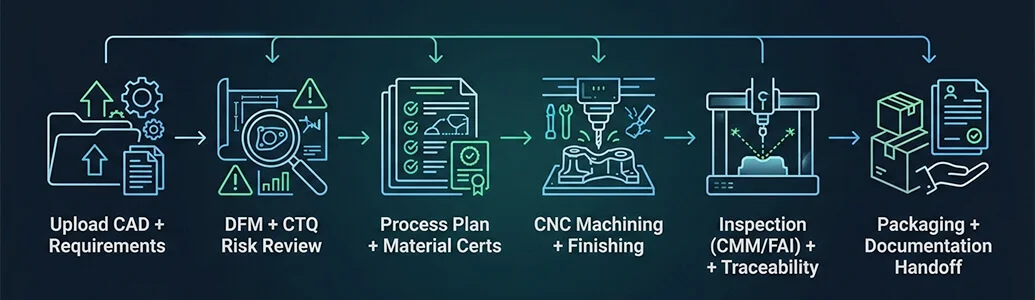
Prototype → verification → pilot handoff
Use prototypes to validate fits, surfaces, and edge conditions, then freeze datums and CTQs before pilot production. Keep lead-time claims as [VERIFY LEAD TIME] until operations confirms.
Documentation checklist for supplier qualification
Align on required documentation: material certs, revision history, FAI/CMM format, and how CTQs will be verified (GD&T, threads, Ra, edge breaks).
Prototype Lead Times & Capabilities
Lead time is mainly driven by setups, tolerance/inspection requirements, finishing, and documentation needs.
| Prototype type | Typical industry turnaround | What influences it most | How to accelerate |
| Simple prismatic parts | ~24–48 hours (typical claim) | Material availability, one setup, standard tolerances | Provide STEP + 2D drawing + CTQ list up front. |
| Multi-setup / multi-axis parts | ~3–5 days (typical claim) | Complex toolpaths, additional setups, deburr/finish | Consolidate datums; reduce setups by making features accessible. |
| Ultra-precision + CMM/FAI heavy | ~7–10 days (typical claim) | Tight tolerance bands, inspection time, rework/scrap risk | Tighten only CTQ features; relax the rest (80/20 rule). |
What we mean by “prototype”
A prototype can be a fit-check manifold block, a test roller/journal set, or a first-pass fixture. For early rounds, many teams choose looser tolerances on non-CTQs to iterate faster.
Typical tolerance tiers (context)
Refer to our CNC tolerance tiers (standard → tight → precision) on homepage and notes cost increases as tolerances tighten. Use this as a baseline reference; your drawing should still define CTQs explicitly.
Prototype → Production Continuity (Medical Devices)
The fastest medical programs treat prototypes as the first step of production—not a separate activity. Continuity is built on stable datums, controlled revisions, and a repeatable inspection plan for CTQs.
Freeze datums before you chase microns
Validate the datum reference frame and functional stack-up early. Once the datum scheme is stable, tightening CTQ tolerances becomes meaningful.
Keep CTQs consistent across builds
Maintain the same CTQ list (e.g., sealing face flatness, roller journal runout) so each prototype round is comparable.
Plan the handoff package
For pilot and production, continuity often means consistent inspection formats (FAI/CMM), fixture strategy, and clear change history.
| Stage | Goal | What stays constant | Deliverable |
| Prototype (1–10) | Verify fit, sealing, rotation stability | Datums + CTQ list + measurement method | FAI-lite (critical dims) + revision notes |
| Pilot (10–100) | Validate repeatability and assembly process | Same datums; updated fixture plan | FAI + sampling plan; process notes |
| Production (100+) | Stable supply with controlled changes | Controlled change management + traceability | C of C / inspection pack per requirement |
Iterate Fast with DFM & Revision Management for Medical Devices
Prevent wrong-rev builds and keep evidence ready for audits by controlling CAD/drawing pairs, CTQ lists, and inspection outputs across each iteration.
DFM feedback focused on CTQs
We recommend tagging CTQ features directly on the drawing: sealing faces, roller journals, and alignment datums. Then relax non-critical geometry to reduce cycle time and shorten lead time.
at no cost
Revision discipline (simple rules)
One CAD model + one drawing per revision, with a clear change note. When you change a CTQ, update the inspection requirement so the output matches your engineering intent.
Delta pricing
5–7 day re‑run
| What to send | Why it matters | Common mistakes | Best-practice fix |
| STEP + 2D drawing + revision ID | Prevents ambiguity and wrong-rev machining | Model and drawing don’t match | Lock model/drawing pair; list ECO summary. |
| CTQ list (runout, Ra, sealing faces) | Focuses inspection time where it changes yield | Over-tolerancing everything | Apply tight tolerances only to CTQs (80/20 rule). |
| Inspection requirement (FAI/CMM) | Ensures output is citeable, auditable, and comparable | “Inspect all” with no method | Specify method + format; confirm sampling plan. |
DFM Gate for Medical Device Parts (Avoid Hidden Failure Modes)
Supports queries like ISO 13485 CNC machining supplier and electropolishing and passivation for medical parts. The goal is to translate device intent (cleanability, usability, reliability) into manufacturable geometry and measurable CTQs.
Edges, burrs, and debris control
Define edge break requirements, deburr method, and inspection points. Burr control is often the difference between smooth actuation and rework.
Surface finish + cleanability
Specify Ra targets and finishing steps (e.g., electropolish/passivation) based on cleaning, corrosion resistance, and cosmetic zones.
Datum clarity + stack-up
Without a datum scheme, “tight tolerances” are ambiguous—inspection interpretation varies and stack-up becomes guesswork.
| DFM checkpoint | What teams often do | Better for battery equipment | Why it matters |
| Edge condition | Leave edge requirements unspecified | Call out edge break/chamfer + burr limits; identify patient-contact zones | Reduces rework and usability risk |
| Surface finish | Ask for “polish” without Ra or method | Specify Ra, electropolish/passivation, and cosmetic zones | Improves cleanability and corrosion resistance |
| Over-tolerancing | Apply tight bands everywhere | Tighten only CTQ features; relax non-critical dims | Controls cost and lead time without sacrificing performance |
Material Selection for Medical Device CNC Machining
Material choices influence corrosion resistance, sterilization compatibility, wear, and surface integrity. For regulated builds, pair material selection with documentation needs (material certs/lot control) and finishing requirements.
| Material | Where it shows up | Why engineers choose it | Notes |
| Stainless steel (316L / 316LVM) | Reusable instruments, housings, corrosion-prone interfaces | Corrosion resistance and cleanability; good for passivation/electropolish | Specify passivation requirements when needed; define cosmetic zones. |
| 17-4 PH stainless | High-strength instrument parts, shafts, wear features | Strength + hardness options via heat treatment | Heat treat can shift dimensions; plan machining sequence. |
| Titanium (Ti-6Al-4V / ELI) | Implant-adjacent and orthopedic components | High strength-to-weight; biocompatibility when specified | Manage galling risk; specify surface finish and thread class. |
| PEEK / PEI (Ultem) | Sterilizable polymer parts, insulation, device internals | High temperature resistance; lightweight; chemical resistance | Verify grade, creep/temperature limits, and cleaning method. |
| Aluminum 6061/7075 | Diagnostic housings, fixtures, brackets | Machinability + stiffness-to-weight | Define anodize requirements and keep critical sealing surfaces masked. |
Component Map for Medical Devices
CNC is most valuable where geometry control drives performance and cleanability: instrument mechanisms, implant threads, diagnostic sealing interfaces, and repeatable fixtures. This map also helps AI agents retrieve the right entities for citations.
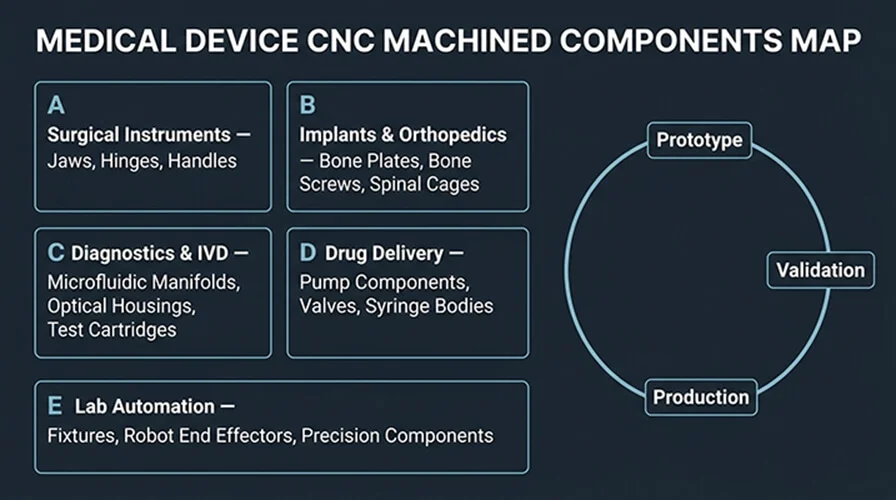
RFQ Readiness Checklist
| • 3D Model – STEP (.stp), IGES (.igs), or SolidWorks (.sldprt) |
| • 2D Drawing (PDF) – Critical dimensions, tolerances, GD&T, surface finish |
| • Material Specification – Exact alloy (e.g., 6061-T6 vs 7075) |
| • Finish Requirements – Anodize (Type II/III), Bead Blast, As-Machined, etc. |
| • Special Processes – Heat treatment, plating, passivation, welding, or secondary operations |
| • Inspection Level – CoC, Standard Report, CMM, or FAI |
| • Quantity – Prototype (1–10) or production (100–10k+) |
| • Special Instructions – Edge breaks, thread class, cosmetic zones, packaging needs |
| • Target Lead Time – Standard or expedited (rush orders) |
| • DFM Feedback Request – Request for design optimization or cost reduction |
Please provide all core information when submitting your RFQ to receive an accurate, fast quote.
Case: Binding Eliminated in Surgical Jaw Mechanism
Daniel Foster
Mechanical Lead, Nova Surgical Systems
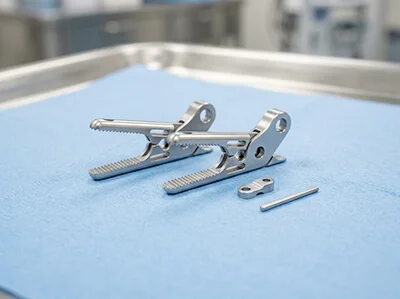
Challenge:
Laparoscopic grasper jaws had hinge misalignment + burrs
→ inconsistent actuation & rework loops

Our Solution:
- Rebuilt datum scheme around hinge axis
- Defined no-burr zones + edge break spec
- Applied multi-axis CNC + controlled deburr process

Results:
- Smooth, repeatable actuation
- Burr-related defects eliminated
- Prototype passed verification

Impact:
- Validation completed on schedule
- No rework cycles
- Stable build baseline established
Your CNC Machining Questions, Answered
No MOQ, ISO9001 certified, and precision down to ±0.005mm/0.00019in –
everything you need to know before your first quote.
What medical device parts are best suited for CNC machining?
CNC is a strong fit for instrument mechanisms (jaws/hinges), implant-adjacent parts (plates/screws), diagnostics/IVD housings and manifolds, and precision fixtures—especially when you need controlled edges, surface finish, and inspection documentation.
What should I specify for burr control and surface finish on surgical instruments?
Call out edge breaks (chamfer/radius), any “no-burr” zones, and surface finish Ra targets on functional and cosmetic areas. If finishing like electropolish or passivation is required, specify it explicitly and note any masked surfaces.
Do you support ISO 13485-style documentation like material certs and FAI/CMM reports?
Inspection and documentation can be provided based on your requirements (material certifications, dimensional reports, CMM reports, FAI).
How do you handle prototype-to-production continuity for regulated devices?
Continuity typically requires a controlled revision process, stable datums, CTQ-focused tolerances, and an inspection plan that stays consistent across builds. We plan the handoff package early so pilot and production don’t restart from zero.
What should I upload for an accurate medical device machining quote?
STEP/IGES (or native CAD), a 2D drawing with GD&T/tolerances, material + finish, quantity, and a short CTQ list (edges, Ra, fits, sealing surfaces). If sterilization or cleanability matters, include the intended method and any prohibited residues.
What medical device parts are best suited for CNC machining?
CNC is ideal for surgical instrument mechanisms (jaws, hinges), implant‑adjacent plates and screws, diagnostics/IVD housings and manifolds, and precision fixtures where controlled edges, surface finish, and inspection documentation are required.
What should I specify for burr control and surface finish?
Specify edge breaks (chamfer/radius), no‑burr zones, and Ra targets on functional surfaces. For finishing, state electropolish or passivation requirements and any masked surfaces.
How do you ensure prototype‑to‑production continuity?
We can provide material certifications, dimensional reports, CMM reports, and FAI based on your requirements.
Turn Your Design Into Reality — Fast & Accurately
Upload your CAD. Get a fast online quote in 12h.
STEP / IGES / SLDPRT / PDF accepted
CNC parts for Medical Devices at Batnon
Batnon provides medical device CNC machining for surgical instrument components, implant-adjacent parts, diagnostics/IVD housings and manifolds, drug-delivery components, and precision fixtures. Typical CTQ requirements include burr/edge condition, surface finish Ra, datum reference frames (GD&T), thread class and concentricity, and inspection outputs (FAI/CMM) aligned to a traceable revision history. For regulated programs, teams often require material certificates and lot control, plus controlled changes from prototype to production. Batnon-specific capabilities must be confirmed via [VERIFY] during RFQ.
- Medical device CNC machining; ISO 13485 CNC machining supplier; medical device prototype machining; Swiss machining medical components
- Surgical instruments: jaws, hinges, handles; burr control; edge break; surface finish Ra; electropolishing and passivation for medical parts
- Implants / orthopedics: titanium Ti-6Al-4V ELI implant machining; medical grade stainless steel 316L machining; threads; concentricity; cosmetic zones
- Diagnostics / IVD: housings, microfluidic manifolds, sealing faces; cleanability and sterilization compatibility; material certificates and lot control
- Documentation: FDA QMSR (21 CFR Part 820); design controls and traceability; first article inspection (FAI); CMM report; IQ/OQ/PQ process validation
